PD Dr. T. Sottmann
Leider hat uns Herr Sottmann in Richtung Stuttgart verlassen.
Publikationen
Eine aktuelle Liste der Publikationen findet sich hier.
CV
Thomas Sottmann studierte Physik an der Universität Göttingen und spezialisierte sich am Max-Planck-Institut für Biophysikalische Chemie auf die Physikalische Chemie. Dort schloss er 1997 seine Doktorarbeit ab. Nach einem Jahr als Post-Doc wechselte er zur Universität zu Köln an das Institut für Physikalische Chemie. Im Jahr 2006 war er Gast-Professor am Department of Chemical Engineering and Materials Science (CEMS) an der Universität Minnesota. Er hält 3 Patente.
Selbstorganisierende amphiphile Systeme
Oberflächenaktive Moleküle sind die Schlüsselkomponenten vieler physikalischer, chemischer und biologischer Systeme. Im letzten Fall bilden Phospholipide die Membran biologischer Zellen und stellen damit 80 % der trockenen Masse lebender Zellen dar. Es ist bekannt, dass oberflächenaktive Moleküle sich in einer großen Vielfalt von Strukturen in Lösungen selbst organisieren. Die treibende Kraft für diese Strukturbildungen ist die Amphiphilie dieser Moleküle. In unserer Gruppe werden die thermodynamischen, strukturellen- und Grenzflächen-Eigenschaften dieser Komplexen Fluide auch im Hinblick auf spätere technische Anwendungen charakterisiert.
Mikroemulsionen als Templatmaterialien für die Synthese nanostrukturierter Materialien
Komplexe Fluide, wie z. B. Mikroemulsionen sind potentielle Kandidaten für die Synthese nanostrukturierter Polymer-Materialien. Allerdings ist die eins-zu-eins Abbildung der gewünschten Mikroemulsionsstruktur auf das Polymer bis heute eine große Herausforderung. In den meisten Fällen bewirkt die Änderung des Monomer/Polymer-Verhältnisses während der Polymerisation eine Phasenseparation. Um die Kinetik des Phasenverhaltens zu verlangsamen formulieren wir hochviskose polymerisierbare Mikroemulsionen, in der die hydrophile sub-Phase durch eine konzentrierte Zuckerlösung ersetzt ist, (ähnlich dem Ansatz von C. Co, J. Am. Chem. Soc. 126, 12746 (2004)). Erste Experimente, die in enger Zusammenarbeit mit der Gruppe von H.T. Davis (CEMS, University of Minnesota) stattfanden deuten auf eine starke Korrelation der Größe des Polymermaterials mit der Größe der zugrunde liegenden Mikroemulsion hin.
Eigenschaften Überkritischer Mikroemulsionen
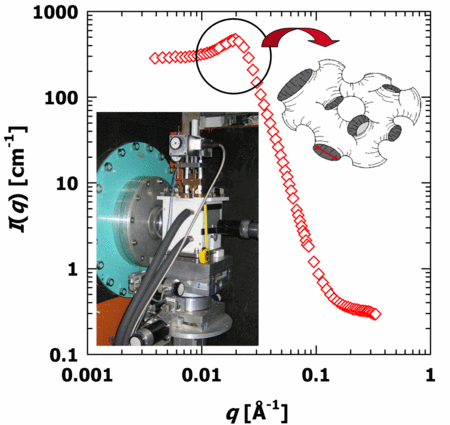
Mischungen aus Wasser und überkritischem Kohlenstoffdioxid (scCO2) gewinnen immer mehr an Bedeutung als unweltfreundliche Lösemittel. Mit bestimmten Tensiden ist es möglich einphasige Mikroemulsionen bestehend aus Wasser, Kohlenstoffdioxid und Tensid zu formulieren. Nach dem Prinzip der überkritischen Mikroemulsions-Expansion (POSME, German Patent DE 102 60 815 B4) können kostengünstige, nanostrukturierte hochporöse Materialen durch die Expansion dieser nahe- und überkritischen Mikroemulsionen erhalten werden. Selbst entwickelte Druckzellen ermöglichen es uns das Phasenverhalten und die Mikrostruktur (mit DLS und SANS) zu bestimmen. Zusätzlich kann man mit Hilfe von Neutronen Spin-Echo (NSE) Experimenten die Membranfluktuationen bikontinuierlicher scCO2-Wasser-Mikroemulsionen untersuchen.
 |
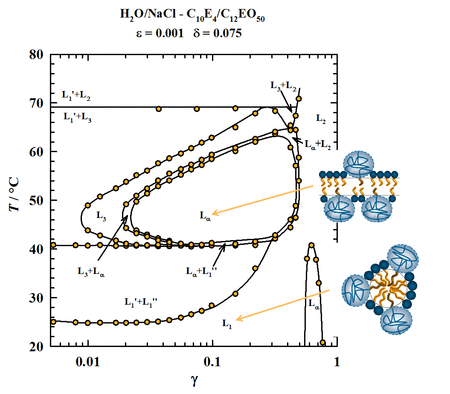
Der Einfluss von Sticker-Polymeren auf Doppelschichtmembranen
Die Rolle von Gast-Molekülen wie kolloidalen Partikeln, Proteinen, DNS oder synthetischen Polymeren in Doppelschichtmebranen sind für biologische Prozesse von großer Relevanz. Um den Einfluss dieser Moleküle auf die Membraneigenschaften zu untersuchen, wurde der Einfluss von Sticker-Polymeren (synthetisiert in der Gruppe von Prof. D. Richter, NoE "SOFTCOMP" (NMP3-CT-2004-502235)) auf Tensid-Doppelschichten untersucht. Erste Experimente zeigen, dass der Austausch von schon kleinen Mengen Tensid durch Polymer sowohl die isotrope Schwammphase L3 als auch die anisotrope lamellare Phase Lα beträchtlich stabilisieren. Kleinwinkel-Neutronenstreuung (SANS), Kleinwinkel-Röntgenstreuung (SAXS) und statische Lichtstreuung in Kombination mit der systematischen Untersuchung des Phasenverhaltens sollen die Rolle des Sticker-Polymers aufklären.
 |
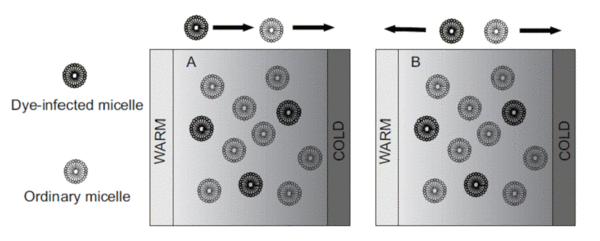
Soret Effekt in Tensid-Systemen und Mikroemulsionen
Thermische Diffusion (der Soret Effekt) beschreibt die Wanderung von Molekülen innerhalb eines Temperaturgradienten und ist ein noch ungelöstes Problem in der Physikalischen Chemie. Viele Experimente zeigen, dass der Effekt stark mit der Grenzfläche zwischen Lösemittel und gelösten Stoff zusammenhängt. Allerdings ist die Grenzflächenspannung zwischen dem Partikel und dem Lösemittel in kolloidalen Modell-Systemen schwierig zu messen. Im Gegensatz dazu kann die Grenzflächenspannung eines Tropfens in einem Mikroemulsionssystem kontrolliert werden, indem man die Krümmung des amphiphilen Films ändert. In Zusammenarbeit mit der Gruppe von S. Wiegand (FZ-Jülich) haben wir im Rahmen eines DFG-Projekts (SO 913, Wi 1684) grade begonnen den Einfluss der Grenzflächenspannung auf den Soret Koeffizienten in Öl-in-Wasser Mikroemulsionen zu untersuchen.
 |